“Iron bacteria are those bacteria that can accumulate iron from their environment.”
These bacteria deposit iron and store iron as storage granules. These bacteria cause iron to react with oxygen and form rust. They form sticky slime layers that allow them to attach and grow. These bacteria oxidize dissolved ferrous in the surroundings and use energy to survive and grow in the environment.
Examples:
Thiobacillus ferrooxidans, Leptospirillum ferrooxidans.
Habitat:
These bacteria live in
- Soil
- Water surfaces
- Groundwater
Characteristics:
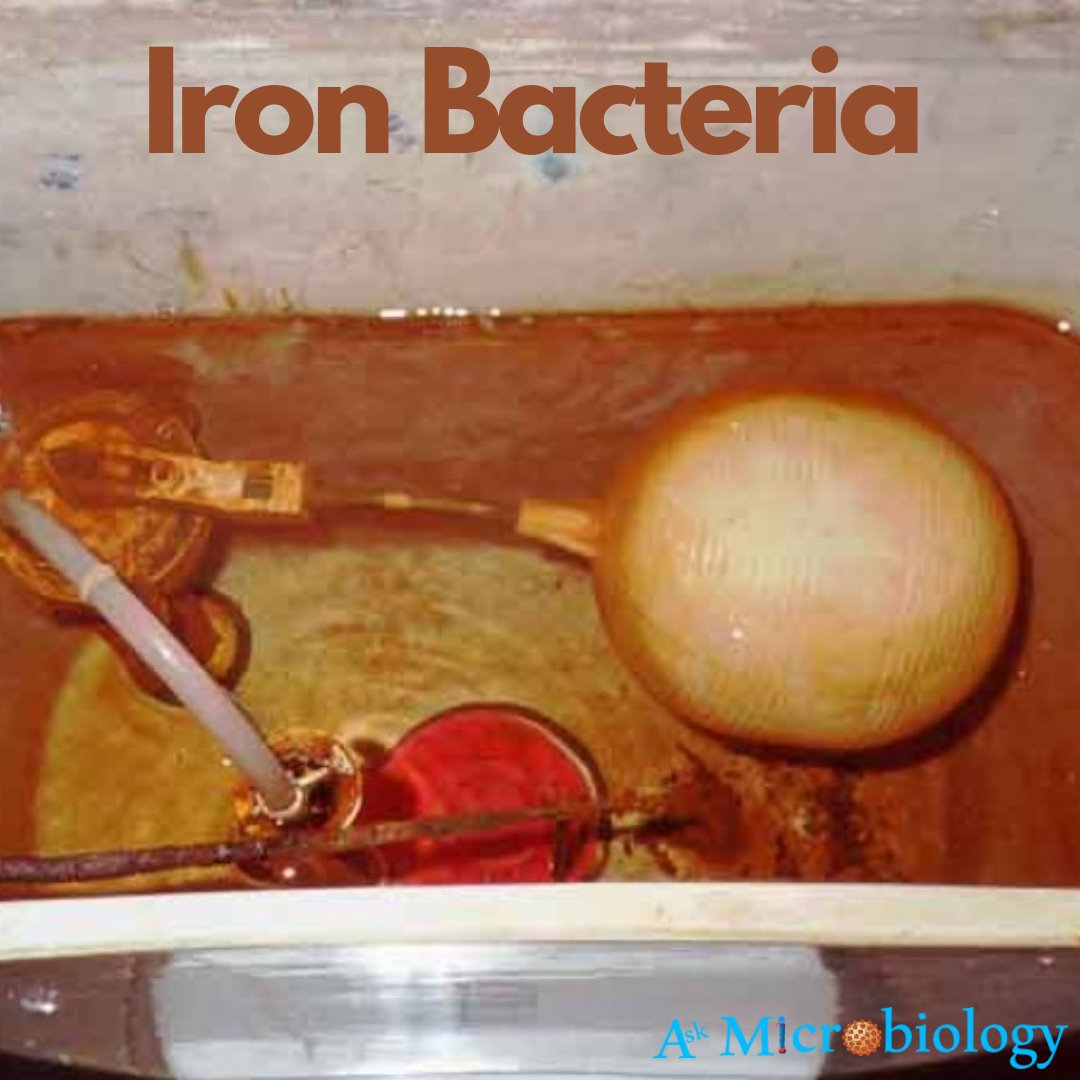
These bacteria live in an anaerobic environment. They use iron as their energy source. These bacteria produce rust colour due to the formation of ferric oxide. When rust is produced, a brown slime layer is formed that helps us to identify the bacteria. These bacteria also produce stingy odour like cucumber, sewage water.
How to culture these bacteria?
Sample must be taken from the soil, underground water and water surfaces in which rust colour is present. Media must contain the dissolved ferric oxide which is utilized by bacteria. These bacteria produce a slime layer or pigmentation during their growth. For identification, gram staining, biochemical test and PCR can be used
Problems caused by these bacteria:
- These bacteria can spoil the drinking quality of water.
- These bacteria produce a stingy smell in the water.
- These bacteria cause yellow, orange colour in the water.
- These bacteria change the taste of water.
Control of bacteria:
These bacteria can be controlled in the water in 3 ways.
- Physical method
- Pasteurization
- Chemical agents
Physical method:
The physical method includes the removal of rust from the groundwater. With the help of proper cleanliness and sanitary conditions, this method can be effective. But it is a time-consuming process.
Pasteurization:
It is done by injecting hot steam of 60ºC for 30 minutes. This is an effective process but very costly
Chemical agents:
Chemicals agent include surfactants that are acids, disinfectants and oxidizing agents.
Surfactants are the chlorine compounds that are most effective to kill iron bacteria
Acids can dissolve iron compounds, destroy bacteria. But there are many precautionary measures to deal with acids.
Oxidizing agents cause oxidation of iron. Iron utilizes from the surroundings and bacteria death occurs.

Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://accounts.binance.info/register-person?ref=IHJUI7TF